Timed practice with TS 10th Class Physical Science Model Papers Set 1 is crucial for improving speed and efficiency during exams.
TS 10th Class Physical Science Model Paper Set 1 with Solutions
Time: 1 Hour 30 minutes
Maximum Marks: 40
General Instructions:
- Read the question paper and understand every question thoroughly and write answers in given 1.30 hrs. time.
- 3 very short answer questions are there in section – I. Each question carries 2 marks. Answer all the questions. Write answer to each question in 3 to 4 sentences.
- 3, short answer questions are there in section – II. Each question carries 3 marks. Answer all the questions. Write answer to each question in 5 to 6 sentences.
- 3 essay type answer questions are there in section – III. Each question carries 5 marks. Answer all the questions. Write answer to each question in 8 to 10 sentences. Internal choice is given in this section.
Part – A (30 Marks)
Section – I (3 × 2 = 6 Marks)
Instructions :
- 3 Very short answer questions are there in section – I.
- Answer ALL the questions. Each question carries 2 marks.
- Write answer to each question in 3 to 4 sentences.
Question 1.
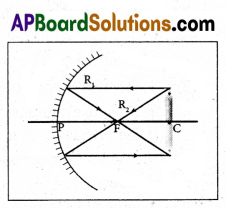
What happens if an object is placed at centre of curvature of a mirror ? Draw the ray diagram.
Answer:
From the ray diagram we conclude that the image of the object will be formed at the same distance as the object and it will be inverted and of the same size. The image is real because it forms on a screen.
Question 2.
What is “Atmospheric refraction” ?
Answer:
- When the light rays pass through the atmosphere having layers of different densities and refractive indices, then refraction of light takes place.
- This refraction of light by the earth’s atmosphere is called “atmospheric refraction”.
Question 3.
What is the role of washing in enriching the ore ?
Answer:
- Washing is one of the physical methods in enriching the ore.
- Ore particles are crushed and kept on a sloppy surface. They are washed with controlled flow of water.
- Less densive impurities are carried away by water flow leaving the more dense ore particles behind.
Section – II (3 × 3 = 9 Marks)
Instructions :
- 3 Short answer questions are there in section – II.
- Answer ALL the questions. Each question carries 3 marks.
- Write answer to each question in 5 to 6 sentences.
![]()
Question 4.
The element oxygen belongs to ? Which group is called as so ? Why ?
Answer:
- The element oxygen belongs to chalcogen family.
- VIA group is called as chalcogen family.
- Because the elements in VIA group form ores with metals, like metal oxides, metal sulphides etc.
- Chalcogenous means ore product. So VIA group is called as chalcogen family.
Question 5.
Generally which group elements form anions. Why ?
Answer:
- V A, VI A, VII A group elements form anions. They are mostly non metal elements.
- To attain eight electrons in the outermost shell, they gain 3, 2, or 1 electron respectively and form anions.
- E.g : 3p – 2, 8, 5 ; p3 – 2, 8, 8
Question 6.
Write the molecular formula of the first four compounds of the homologous series of aldehydes.
Answer:
Homologous series of aldehydes are Formaldehyde, Acetaldehyde, Pro-pionaldehyde and Butylaldehyde.

Section – III (3 × 5 = 15 Marks)
Instructions :
- 3 Essay answer questions are there in section.
- Answer ALL the questions. Each question carries 5. marks.
- Internal choice is given in this section.
- Write answer to each question in 8 to 10 sentences.
Question 7.
Why does respiration considered as an exothermic reaction ? Explain.
(OR)
How do you prepare washing soda ? What are its uses ?
Answer:
- We need energy to stay alive.
- We get this energy from food we eat.
- During digestion, food is broken down into simpler substances.
- For example, rice and potato contains starch.
- The starch breaks down to form glucose.
- This glucose combines with oxygen in the cells of our body and releases energy, which helps to do the various works.
- During this process, energy is given out. Hence this reaction can be called exothermic reaction.
- The special name of this reaction is respiration;
- So, respiration is considered as exothermic reaction.
- C6H12O6 + 6O2 → 6CO2 + 6H2O + Q (Energy)
(OR)
Preparation of washing soda :
- Washing soda can be obtained from sodium chloride.
- Baking soda is obtained from sodium chloride as follows.
NaCl + H2O +CO2 + NH3 → NH4Cl + NaHCO3
- On heating,faking soda produces sodium carbonate.
2NaHCO3 Na2CO3 + H2O + CO2
Na2CO3 + H2O + CO2
- Recrystallization of sodium carbonate gives washing soda.
Na2CO3 → 10H2O + Na2CO3.10H2O
Uses of washing soda:
- Washing soda (sodium carbonate) is used in glass, soap and paper industries.
- It is used in the manufacture of sodium compounds such as borax;
- Sodium carbonate can be used as a cleaning agent for domestic purposes.
- It is used for removing permanent hardness of water.
Question 8.
Write characteristics of image foraled due to convex lens at various distances.
(OR)
Heisenberg contradicts Neils Bohr. Explain in what way he contradicts.
Answer:
| Position of the object | Position of the image | Relative size of the image | Nature of the image |
| 1) At infinity | Focal point | Highly diminished | Real and inverted |
| 2)Beyond centre of curvature (C2) | Between F1 and C1 | Point size diminished | Real and inverted |
| 3)At the centre of curvature (C2) | At C1 | Same size | Real, inverted |
| 4)Between centre of curvature focal point (C2 and F2) | Beyond centre of curvature | Magnified | Real, inverted |
| 5) At focal point (F2) | At infinity | Highly magnified | Real and inverted |
| 6) Between focal point and pole F2 and P | On the same side of the lens of the object | Magnified | Virtual and erect |
(OR)
- According to Bohr electrons revolve around nucleus in definite paths or orbits, so the exact position of the electron at various times will be known to us.
- In order to explain Bohr’s postulate we have to know the velocity and exact position of electron.
- In order to find the position of electron we have to take the help of suitable light to find the
position. As the electrons are very small light of very short wavelength is required for this task. - This short wavelength light interacts with the electron and disturbs the motion of the electron.
- Hence, it is not possible to find the exact position and velocity of electron simultaneously. This was stated by Heisenberg which is called Heisenberg’s principle or uncertainty.
- In this way Heisenberg contradicts Neils Bohr.
![]()
Question 9.
Derive an expression to find drift velocity of electrons.
(OR)
a) State Right hand rule with a labelled diagram.
b) A coil of insulated copper wire is connected to a Galvanometer. What happens, if a bar magnet is
i) pushed into the coil ?
ii) withdrawn from inside the coil?
iii) held stationary inside the coil ?
Answer:
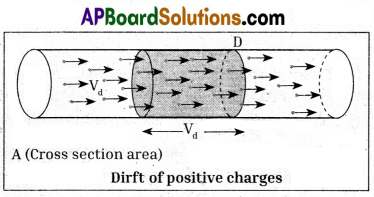
- Consider a conductor with cross-sectional area A. Assume that the two ends of the conductor are connected to a battery to make the current flow through it.
- Let ‘Vd’ be the drift speed of the charges and ‘n’ be the number of charges.present in the conductor in unit volume.
- The distance covered by each charge in one second is ‘Vd’.
- Then the volume of the conductor for this distance = AVd
- The number of charges contained in that volume = n.AVd
- Let q be the charge of each carrier.
- Then the total charges crossing the cross-sectional area at position D in one second is ‘nq AVd’.
This is equal to electric current.
∴ Electric current I = nq AVd.
∴ Vd = \(\frac{\mathrm{I}}{\mathrm{nqA}}\)
(OR)
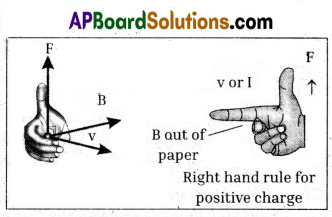
i) Flemming right hand rule indicates the direction of magnetic force acting on a moving charge.
ii) It is used when velocity and field are perpendicular to each other. If the fore finger points towards the direction of velocity of charge or current (I), middle finger points to the direction of field (B), then thumb gives the direction of force (F).
b)
i) Needle in Galvanometer gets deflected. (Or) Current is generated in the coil.
ii) Needle in Galvanometer gets deflected. (Or) Current is generated in the coil.
iii) Needle in Galvanometer does not get deflected. (Or) Current is not generated in the coil.
Part – B (10 × 1 = 10 Marks)
Instructions :
- Answer ALL the questions.
- Each question carries 1 mark.
- In this section there are 4 options (A / B / C / D) to each question. Choose the appropriate answer and write the answer in the brackets given against the question. Part – B must be attached to the answer booklet of Part – A.
Question 1.
The ratio of the focal length of spherical mirror to its radius of curvature is
A) 0.5
B) 1
C) 2
D) 3
Answer:
A) 0.5
Question 2.
Ammonia is formed by the reaction of gases
A) H2 and O2
B) N2 and O2
C) N2 and H2
D) H2 and He
Answer:
C) N2 and H2
Question 3.
WhichofthefoBowingisthemostaccuratewayofshowingneutralization?
A) Acid + Base → (Acid – base solution
B) Acid + Base → (Salt + Water
C) Acid + Base → (Sodium chloride + Hydrogen
D) Acid + Base → (Neutral solution
Answer:
B) Acid + Base → (Salt + Water
Question 4.
Which of the following is used to enhance the taste of food?
A) sodium carbonate
B) sodium sulphate
C) sodium chloride
D) sodium bicarbonate
Answer:
B) sodium sulphate
Question 5.
Which of the following is obtained by recrystaihzation of sodium carbonate ?
A) Washing soda
B) Baking soda
C) Bleaching powder
D) None of these
Answer:
A) Washing soda
Question 6.
The focal length of a lens depends on which of the following factors.
A) Radius of curvature
B) Material of the lens
C) Medium in which the lens is placed
D) All the above
Answer:
D) All the above
![]()
Question 7.
At where the object is placed, the image formed at infinity
A) at C2
B) at F2
C) between C2 and F2
D) None
Answer:
A) at C2
Question 8.
Red colour of the Sun at the time of sunrise and sunset is because of
A) red colour is least scattered
B) blue colour is least scattered
C) all colours are equally scattered
D) red colour is scattered the most
Answer:
A) red colour is least scattered
Question 9.
The following is metalloid
A) Silicon
B) Na
C) Carbon
D) Fe
Answer:
A) Silicon
Question 10.
Which of the following metals are moderately reactive?
A) Pb
B) Fe
C) Zn
D) All of the these
Answer:
D) All of the these