These AP 7th Class Science Important Questions 2nd Lesson Acids and Bases will help students prepare well for the exams.
AP State Syllabus 7th Class Physical Science 2nd Lesson Important Questions and Answers Acids and Bases
7th Class Science 2nd Lesson Acids and Bases Important Questions and Answers
Question 1.
Write the names of food substances that you know in the appropriate column, based on their taste.
Answer:
| Sweet | Sour | Bitter | Salty | Spicy |
| Sugar | Lemon Juice | Bittergourd | Common Salt | Red Mirchi |
| Custard apple |
Apple Juice | Bittergouard Snakegourd |
Sea water | Chillies |
| Jack fruit | Citron Juice | Soapnut | Ground water | Pepper |
| Mango | Grapes Juice | Bitter Almond | Mixture | Green Chillies |
| Date fruit | Orange Juice | Bitter Ribbedgourds | Rasam | Bar of Cloves |
| Papaya | Gooseberry | Bitter Cucumber | Iodised salt | Samosa |
![]()
Question 2.
Take solutions of some juices of fruits, vegetables, cold drinks and various solutions and test them with blue and red litmus papers. List out results in the table, by marking (S) in the relevant column.
Answer:

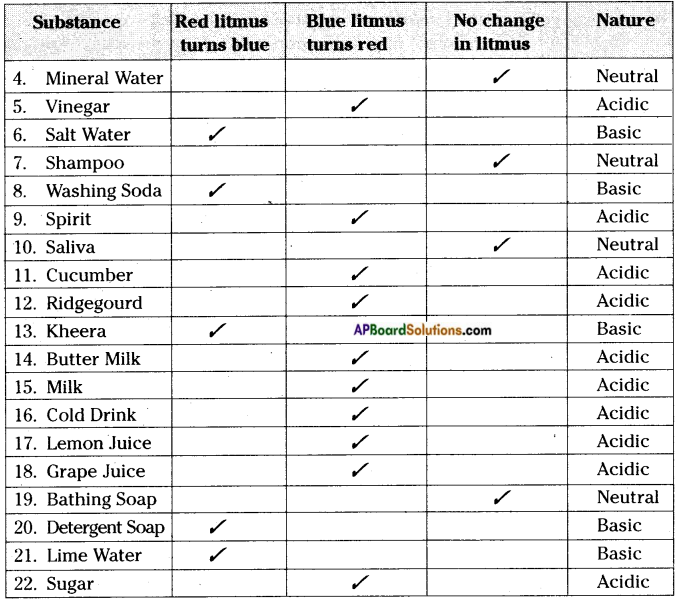
Question 3.
How are hydroxides prepared? What is their nature?
Answer:
- Oxides of Magnesium when dissolved in water form Magnesium Hydroxide.
- Similarly oxides of Potassium and Sodium form Potassium Hydroxide and Sodium Hydroxide when dissolved in water.
- All these are basic in nature.
- They would be soapy when touched.
![]()
Question 4.
Classify the substances given in the table under question 2 into the following categories. Acidic substances, Basic substances, and Neutral substances.
Answer:
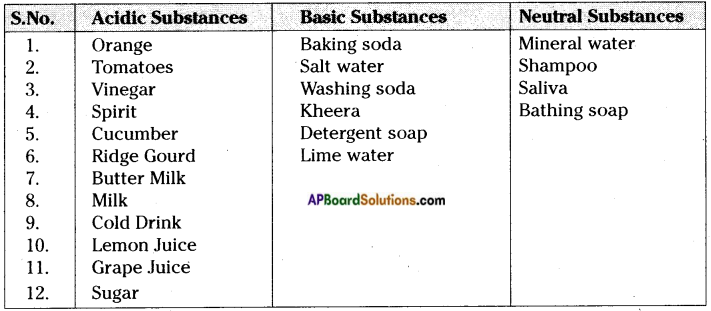
Question 5.
When do you get a neutral solution from acids and bases?
Answer:
When acids and bases are mixed in definite proportions they give a neutral solution.
Question 6.
How are salts produced? Give an example.
Answer:
- We have seen in the process of neutralization, both the acidic and the basic qualities are destroyed.
- Actually when an acid and a base are mixed, a chemical reaction takes place and salts are produced.
- For example by neutralizing Hydrochloric acid solution with Caustic soda (Sodium Hydroxide) solution, a salt Sodium Chloride is produced.
- Production of salt depends on the quantity of acid and base by proportion in a very specific manner.
Question 7.
Write the uses of acids, bases and salts.
Answer:
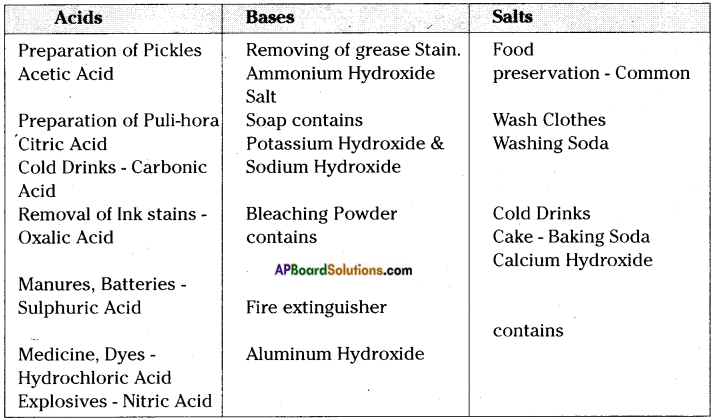
Question 8.
What are indicators? How are they useful?
Answer:
- The paper strips like litmus paper strips change colour based on the nature of the material put on them.
- These are all indicators.
- They tell us whether a substance is acidic or basic.
- They are used extensively in studying substances and their properties.
![]()
Question 9.
Are there any acids apart from natural acids? What are they?
Answer:
Apart from natural acids, there are certain acids like Hydrochloric acid, Sulphuric acid and Nitric acid etc., that are prepared from minerals.
Question 10.
You might have touched soap water or lime water. How do you feel? What is your conclusion?
Answer:
- When I have touched soap water and lime water, they are soapy to touch.
- These substances are basic in nature.
Question 11.
Who discovered Hydrogen? What is its colour?
Answer:
- Henry Cavendish, an Italian Scientist.
- He discovered a colourless gas called Hydrogen.
Question 12.
Write the properties of acids and bases from your observations with the experiments done by you.
Answer:
| Acids | Bases |
| 1) Acids are sour to taste.
2) They react with bases forming salts. 3) These turn blue litmus red. |
1) Bases are bitter to taste.
2) They react with acids forming salts. 3) These turn red litmus blue. |
Question 13.
Are all neutral solutions salts? Give examples.
Answer:
- All neutral solutions are not salt solutions.
- Sugar or Starches are neutral but they are not salts.
![]()
Question 14.
How do you draw a red flower using indicators to get the desired colour?
Answer:
- We should take some turmeric powder. Add a bit of water to it and prepare turmeric paste.
- We should rub the turmeric paste on a white paper.
- And draw a flower on that paper with a pencil.
- Colour the flower with soap water using a brush.
- We notice that the colour of the flower changes to red.
- Similarly take some lime water.
- Add a few drops of it on a piece of turmeric paper.
- The colour changes to red.
Question 15.
Turmeric paper is a natural indicator, along with turmeric indicator paper, we can also prepare other natural indicators mentioned in the table below. Carry on tests and note your observations.
Answer:
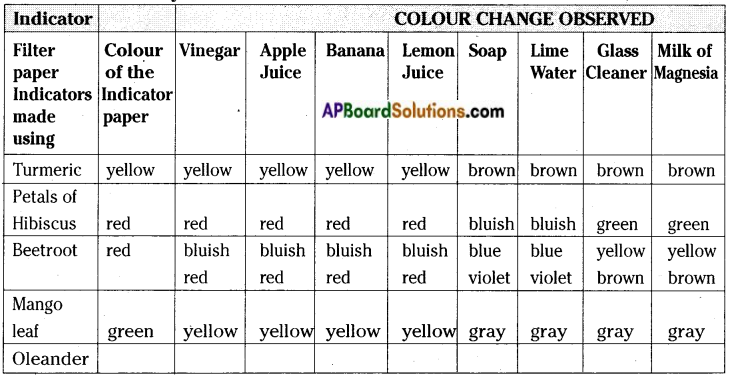
Question 16.
How can you prepare a base by doing a simple experiment?
Answer:
- We should burn a small piece of Magnesium ribbon by holding it with tongs.
- Collect the white ash and dissolve it in a little water.

- Touch the solutions formed and test this solution with blue litmus and red litmus.
- This solution turns red litmus blue.
- Oxides of Magnesium when dissolved in water form Magnesium Hydroxide.
- Magnesium Hydroxide is a base.
Question 17.
Take the following substances and test them with Phenolphthalein and Methyl orange solutions. Draw a table and record your observations. Substances Orange, Tomatoes, Baking soda, Mineral water, Vinegar, Saltwater, Shampoo, Washing soda.
Answer:
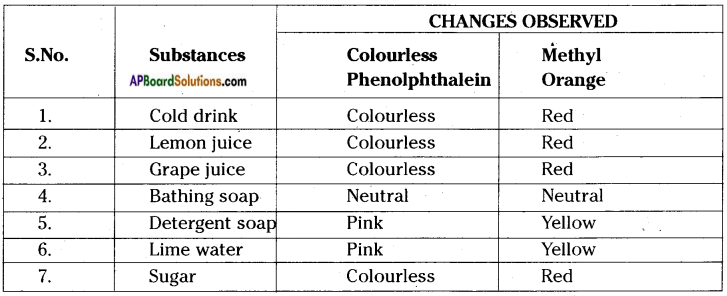
Question 18.
What changes in colour take place in the indicators Phenolphthalein and Methyl Orange when you test with the following substances. Spirit, Saliva, Cucumber, Ridge gourd, Kheera, Buttermilk, Milk.
Answer:
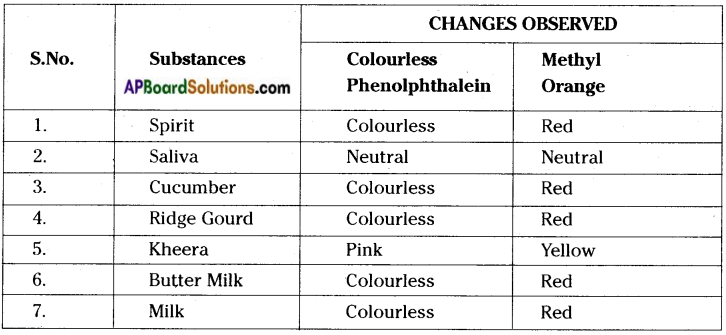
![]()
Question 19.
Record your observations when you test the following substances with the indicators Phenolphthalein and Methyl Orange.
Substances: Cold drink, Lemon Juice, Grape Juice, Bathing soap, detergent soap, Lime water and Sugar.
Answer:
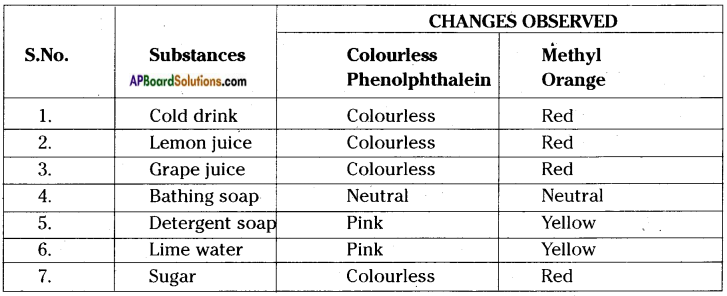
Question 20.
How can you prepare Hydrogen in the laboratory by a simple experiment? With which test do you confirm the presence of Hydrogen?
Answer:
- Take a natural acid say lemon juice in six test tubes and add Copper, Zinc, Magnesium, Iron, Brass, Aluminium pieces to each one of the test tubes separately.
- Note the observations.
- Light a matchstick and introduce it into the test tubes.
- The gas releasing catches fire with a sound.
- This gas is Hydrogen. This is the test for the presence of Hydrogen.
Question 21.
How do you prepare carbondioxide? What substances do you need for its preparation? How is it tested?
Answer:
Preparation of carbondioxide:
- Take lemon juice in two test tubes and add some pieces of marble to one test tube and egg shells to another.
- Bring a burning match stick near the test tube.

- The flame of the burning match stick is put off.
- This is due to release of carbon dioxide gas.
- Pass the gas into lime water. Lime water turns milky white.
- This is the test for carbondioxide.
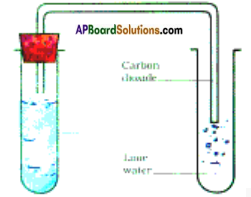
- Lemon juice and pieces of marble or egg shells are the substances needed for the preparation of carbondioxide.
Question 22.
When is the solution made by mixing an acid and a base, a neutral solution? Describe a small activity.
Answer:
- Take a clean test tube using a clean dropper and put 10 drops of Hydrochloric acid carefully in it and also add 2 drops of phenolphthalein indicator solution.
- The solution becomes colourless as the indicator phenolphthalein is colourless in acidic medium.
- Now add Caustic soda solution (whose concentration is equal to Hydrochloric acid) drop by drop to the test tube.
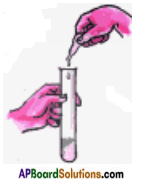
- After each drop shake the test tube well and see if there is any change in the colour of the solution.
- Keep adding the Caustic soda drop by drop until the colour begins to turn pink.
- Now the test tube has basic solution.
- Add one drop of Hydrochloric acid and see if it changes back to its original (colourless) state.
- If not, then add one more drop of Hydrochloric acid.
- Keep doing this until the solution in the test tube becomes colourless again.
- Now the solution in the test tube is neutral. It can be tested with litmus paper.
![]()
Question 23.
What are your conclusions from the experiment done in the above question no. 9?
Answer:
- On the basis of the experiment done above, we can conclude that adding excess of base to acidic solution we can turn acidic solution into basic solution.
- If we are given basic solution we can similarly convert it into acidic solution.
- In the above experiment, if we add excess of base (caustic soda) to acidic solution it converts into a base.
- Similarly we can change base into an acid.
Question 24.
Test the following salt substances with red litmus and blue litmus papers. Record your observations in the table.
Answer:
| Salt substance | Effect on Blue litmus paper |
Effect on Red litmus paper |
| Copper sulphate | Turns red | No effect |
| Common salt | No effect | No effect |
| Sodium carbonate | No effect | Turns blue |
Question 25.
Collect some salt substances with the help of your teacher. Make their salt solutions. Test the salt solutions with blue litmus and red litmus papers. Classify these salts and record your observations in the table.
Answer:

Question 26.
What acids are present in the following substances? Collect the information and write. Substances: Vinegar, Lemon or orange, State cheese Buttered milk or curd, Spinach or Tomato.
Answer:
| Acid | Substance |
| Acetic Acid | Vinegar |
| Citric Acid | Lemon, Orange |
| Butyric Acid | Stale cheese |
| Lactic Acid | Battered Milk, Curd |
| Oxalic Acid | Spinach, Tomato |
![]()
Question 27.
You are asked to find out the acids present in the substances, placed in front of you. Write the names of the acids present in them by collecting information from any source available to you.
Substances placed in front of you : Apples, Tea, Olive oil and fats.
Answer:
| Acid | Substance |
| Malic Acid | Apples |
| Tannic Acid | Tea |
| Oleic Acid | Olive Oil |
| Stearic Acid | Fats |
Question 28.
Grapes, Palm oil, Amla (emblica fruit) and Urine are put before you. You are asked to give the information about the acids that are present in those substances. Collect the information and record in a table.
Answer:
| Acid | Substance |
| Tartaric Acid | Grapes, Tamarind |
| Palmtic Acid | Palm Oil |
| Ascorbic Acid | Amla (emblica fruit) |
| Uric Acid | Urine |
Question 29.
Write the names of some substances that contain bases that we use in our daily life.
Answer:
| Substance | Base |
| Lime Water | Calcium Hydroxide |
| Glass Cleaners | Ammonium Hydroxide |
| Soaps | Sodium Hydroxide / Potassium Hydroxide |
| Milk of Magnesium | Magnesium Hydroxide |
Question 30.
Methyl orange and Phenolphthalein are indicators. Collect the information about their colour change in Acid and Basic solutions.
Answer:
Methyl orange gives red colour with acids and yellow colour with bases. Phenol¬phthalein remains colourless in acidic solution while it turns pink in basic solution.
Question 31.
What sort of food do we eat?
Answer:
- We eat food of different tastes.
- The food will be a balanced diet.
Question 32.
What kind of tastes do food substances we eat have?
Answer:
Sweet, Sour, Bitter, Salty and Spicy tastes of food we eat.
Question 33.
Do you find any difference in the taste of a ripe and a raw fruit?
Answer:
- Ripe fruit is sweet to eat.
- Raw fruit is also sweet to eat but is hard relative to ripe fruit.
Question 34.
Do some substances change their taste when cooked?
Answer:
Some substances change their taste when cooked.
Question 35.
Add sugar to lemon juice. What change do you notice in the taste?
Answer:
The lemon juice becomes sweet to taste when sugar is added.
Question 36.
Is there any difference in the taste without sugar?
Answer:
Without sugar lemon juice will be sour to taste.
![]()
Question 37.
Do some substances change colour when added to other substances?
Answer:
Yes. Some substances change colour when added to other substances.
Question 38.
Have you observed any change when lime water is added to turmeric?
Answer:
When lime water is added to turmeric the colour changed to red.
Question 39.
What are indicators?
Answer:
Those substances that indicate a change in colour when some substances are added to them are known as indicators.
Question 40.
How do you prepare lime water? In what way is this useful to you in day to day life?
Answer:
- Take half a beaker full of water. Add about 5 gm of lime that we apply to betel leaves.
- Stir the beaker well and let it stand overnight.
- Filter this solution the following day.
- This filtrate is lime water. This solution should be transparent.
- We use this filterate in our experiments.
- Lime is used when we consume betel leaves.
Question 41.
You are given a substance. What do you do to identify it as an acid, a base or neutral in nature?
Answer:
- The substance that turns blue litmus to red is acidic in nature.
- The substance that is soapy to touch and turns red litmus to blue is basic in nature.
- If the substance given did not change the colour of either blue or red litmus, then it is said to be neutral in nature.
Question 42.
Take things which are sour in taste and used as food. Eg. Curd, lemon juice. If you check these substances with blue and red litmus what happens?
Answer:
- When a drop of curd is put on blue litmus paper the colour changed to red indicating the acidic nature of curd.
- When a drop of lemon juice is put on the blue litmus paper, the colour of the paper changed to red indicating the acidic nature of lemon juice.
Question 43.
Why have you experienced a burning sensation when bitten by an ant?
Answer:
- I have experienced a burning sensation when bitten by an ant.
- The sensation is due to the presence of formic acid that the ant releases.
![]()
Question 44.
How is it possible to make-believe Blood comes out of a lemon?
Answer:
- When a magician or a juggler cuts a lemon, blood flows out of it. Actually, he uses a knife dipped in Methyl Orange or hibiscus solution to cut the lemon.
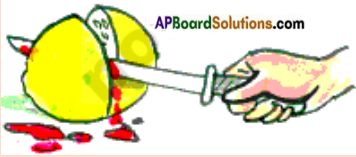
- When Methyl Orange is mixed with citric acid (lemon juice) it turns red.
- But it is not blood.
- In many of our places we find people doing this and presenting it as magic.
Question 43.
Why are the inner sides of vessels made up of brass and copper coated?
Answer:
- When some substances are kept in a copper container for a long time then a blue-green layer is formed in the inner walls of the container.
- Copper reacts with the acids present in the substances and forms a blue-green compound.
- To avoid this reaction the inner walls of these vessels are coated with Tin.
Question 44.
Why are pickles, jams, jellies preserved in glass, porcelain and plastic containers?
Answer:
- The acidic or basic nature of the substances pickles, jams, jellies react with the material of the containers if they are preserved in metallic containers.
- These corrode the metal of the container.
- To avoid this these are preserved in glass, porcelain or plastic containers.
- The material of these containers do not react with the substances preserved.
Question 45.
Ravi said that acid rains are causing damage. Do you agree with his statement?
What are acid rains? Where do you witness such rains in our state?
Answer:
Yes. I agree with his statement because
- Acid rains are the combination of Carbonic acid, Sulphuric acid and Nitric acid with rain water.

- Acid rains cause damage to buildings and to our skin.
- Industrial waste gases contain Sulphur dioxide,
Nitrogen Oxide, Carbon dioxide. - When they get mixed with moisture they change to acids.
- Acid rains are also witnessed in our State in the Visakhapatnam district.
![]()
Question 46.
You are given Hydrochloric acid. How can you make it into a solution which is neither acidic nor basic?
Answer:
- 10 drops of Hydrochloric acid is taken in a clean test tube.
- A drop of methyl orange indicator input is that acid in the test tube.
- The colour of the solution becomes red due to the indicator.
- With the help of a dropper carefully drop drops of caustic soda into the test tube and shake it.
- When an additional drop is put in the solution, the colour changes to yellow, indicating that the solution becomes a base.
- If we add only one drop of Hydrochloric acid in the test tube, the colour of the solution almost disappears indicating that the solution in the test tube is neutral.
Question 47.
You are asked to make the given caustic soda solution into neither acidic nor basic. What would you do?
Answer:
- In a clean test tube 10 drops of caustic soda solution is taken.
- One drop of Methyl orange indicator is added to it.
- The solution turns to yellow colour indicating that the solution is basic in nature.
- With the help of a dropper drops of hydrochloric acid are added to the solution in the test tube.
- By the drop of a drop of Hydrochloric acid in the process, the yellow colour of the solution disappears.
- The solution in the test tube is now neutral in nature.
Question 48.
Why is the use of natural organic manure becoming more preferred?
Answer:
- In recent years the use of chemical fertilizers has increased.
- Though the use of chemical fertilizers increases production it changes the nature of the soil.
- Some fertilizers increase the acidity of the soil and some its basicity.
- Thus use of natural organic manure is becoming more preferred these days.
Question 49.
Why is our sweat like salt?
Answer:
- Our body needs many types of salts.
- We lose some salts through excretion.
- So sweat is salty.
Question 50.
Charitha sprinkled lime water on turmeric idol. It turned into red colour. Guess and write the reasons.
Answer:
Turmeric is a natural indicator. When we sprinkle lime water (calcium hydroxide) which is a base, turmeric indicates red colour in the basic medium.
![]()
Question 51.
If Hibiscus, Turmeric, Rose petals are given to you, mention the use of the above materials for investigating Acids and Bases in the school lab. MiiKiitaa
Answer:
Rose, turmeric and hibiscus are natural indicators. Hence, we can use them to identify the chemical nature of different substances.
Question 52.
a) You have tested the following materials with litmus paper. Fill the table according to that and answer the following questions.

b) In the above table
Acids are: ……………….
Bases are: ……………….
Neutral solutions are: ……………….
Answer:

From the above table
The Acids are – Lemon juice, Grape juice and Orange.
The Bases are – Lime water, Soap water.
The Neutral solutions are – Milk, Sugar solution and Salt water.
Question 53.
Read the following table and answer the questions.

i) Which substances will change blue litmus to red?
ii) Which substances will change red litmus to blue?
iii) Which substances will not be affected by blue litmus or red litmus?
iv)What kind of substances are litmus papers?
Answer:
i) Acids like orange juice, vinegar turn blue litmus red.
ii) Basic substances like baking soda solution turn red litmus blue.
iii) Sugar solution will not be affected by either red litmus or blue litmus.
iv) Litmus papers are called indicators.
![]()
Question 54.
Burn a small piece of Magnesium ribbon by holding it with tongs.

Collect the white ash formed and dissolve it in little water.
Test the solution formed with blue litmus and red litmus.
Now answer the the questions given below,
i) What is the aim of this experiment?
ii) Is the composition of the ash formed different from Magnesium ribbon? If so, how?
iii) What substance is formed when the ash is mixed with water?
iv) What inference can be made on the basis of this experiment?
Answer:
i) To test the effect of basic substances on red litmus paper.
ii) Yes, the substance formed after burning looks like ash.
iii) Magnesium Hydroxide.
iv) Basic substances (or) Magnesium Hydroxide.
Question 55.
Observe the experiment and answer the following questions.
Mix a spoon of vinegar and a pinch of baking soda in a test tube. Pass the gas that is released into another test tube containing lime water as shown here.
i) What is the aim of this experiment?
ii) Name the apparatus used to conduct this experiment.
iii) Name the gas released in the test tube that on mixing vinegar with baking soda.
iv) What is the change observed in the lime water on passing the gas?
Answer:
i) Test tubes, rubber cork, delivery tube.
ii) Vinegar, baking soda, water, lime water.
iii) Carbon dioxide.
iv) Lime water turns milky.
