Students get through AP Inter 2nd Year Chemistry Important Questions 1st Lesson Solid State which are most likely to be asked in the exam.
AP Inter 2nd Year Chemistry Important Questions 1st Lesson Solid State
Very Short Answer Questions
Question 1.
What is meant by the term coordination number ?
Answer:
The number of nearest neighbouring particles of a particle is defined as the co-ordination number.
(Or)
The number of nearest oppositely charged ions surrounding a particular ion is also called as co-ordination number.
E.g.: Co-ordination no. of Na+ in NaCl lattice is ‘6’.
Question 2.
What is the co-ordination number of atoms in a cubic close – pack structure ?
Answer:
The co-ordination number of atoms in a cubic close pack structure is ’12’.
![]()
Question 3.
What is the co-ordination number of atoms in a body – centered cubic structure ?
Answer:
The co-ordination number of atoms in a body – centered cubic structure is ‘8’.
Question 4.
How do you distinguish between crystal lattice and unit cell ? [Board Model Paper]
Answer:
Crystal lattice : A regular arrangement of the constituent particles of a crystal in the three dimensional space is called crystal lattice.
Unit cell: The simple unit of crystal lattice which when repeated again and again gives the entire crystal of a given substance is called unit cell.
Question 5.
What is Schottky defect ? [A.P. IPE 2015]
Answer:
Schottky defect:
- “It is a point defect in which an atom or ion is missing from its normal site in the lattice”.
- In order to maintain electrical neutrality, the number of missing cations and anions are equal.
- This sort of defect occurs mainly in highly ionic compounds, where cationic and anionic sizes are similar.
In such compounds the co-ordination number is high.
Ex.: NaCl, CsCl etc. - Illustration :
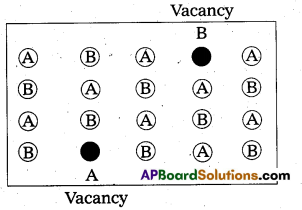
- This defect decreases the density of the substance.
Question 6.
What is Frenkel defect ? [A.P. IPE 2015]
Answer:
Frenkel defect:
- “It is a point defect in which an atom or ion is shifted from its normal lattice position”. The ion or the atom now occupies an interstitial position in the lattice.
- This type of a defect is favoured by a large difference in sizes between the cation and anion. In these compounds co-ordination number is low.
E.g.: Ag – halides, ZnS etc. - Illustration :
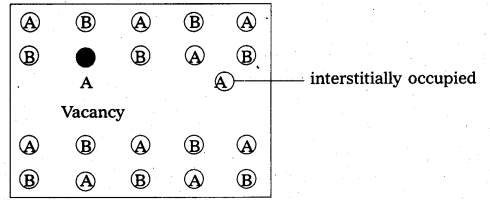
- Frenkel defect do not change the density of the solids significantly.
![]()
Question 7.
What are f – centers ?
Answer:
- f – centers are the anionic sites occupied by unpaired electrons.
- These import colour to crystals. This colour is due to the excitation of electrons when they absorb energy from the visible light.
- f – centres are formed by heating alkyl halide with excess of alkali metal.
E.g. : NaCl crystals heated in presence of Na – vapour, yellow colour is produced due to f – centres.
Question 8.
Why X – rays are needed to probe the crystal structure ?
Answer:
According to the principles of optics, the wavelength of light used to observe an object must be no greater than the twice the length of the object itself. It is impossible to see atom s using even the finest optical microscope. To see the atoms we must use light with a wavelength of approximately 10-10 m. X – rays are present with in this region of electromagnetic spectrum. So X – rays are used to probe crystal structure.
Question 9.
Explain Ferromagnetism with suitable example.
Answer:
Ferromagnetic Substances : Some substances containing more number of unpaired electrons are very strongly attracted by the external magnetic field. In Ferromagnetic substances the magnetic moments in individual atoms are all alligned in the same direction. Such substances are called Ferromagnetic Substances. In ferromagnetic substances the field strength B > > > H.
E.g.: Fe, Co and Ni.
Question 10.
Explain Ferrimagnetisms with suitable example.
Answer:
Ferrimagnetism is observed when the magnetic moments of the domains in the substance are aligned in parallel and anti parallel directions in unequal numbers.
- These are weakly attracted by magnetic field as compared to ferromagnetic substances.
- These lose ferrimagnetism on heating and becomes paramagnetic.
Question 11.
Explain Antiferromagnetism with suitable example.
Answer:
Substances like Mno showing anti-ferromagnetism having domain structure similar to ferromagnetic substance, but their domains are oppositely oriented and cancel out each others magnetic moment.
![]()
Question 12.
What are Tetrahedral voids ?
Answer:
The second layer spheres over the first layer arrangement, the spheres of second layer are placed in the depressions of the first layer. All the triangle voids of the first layer are covered by the spheres of second layer. These are called ‘Tetrahedral voids’.
Question 13.
What are Octahedral voids ?
Answer:
The triangular voids in the second layer are above the triangular voids in the first layer. Such voids are surrounded by six spheres and are called ‘Octahedral voids’.
Question 14.
What are n-type semiconductors ?
Answer:
Silicon and Germanium belong to IVA group and have four valence electrons. When these elements are doped with VA group like P or As which have 5 valence electrons some of lattice sites of Si are replaced by VA group element. Each VA group element forms four bonds with four Si atoms and fifth electron is extra and becomes delocalised. These delocalised electrons increase the conductivity. The increase in conductivity is due to negatively charged electrons. Hence it is called n – type semi conductor.
Question 15.
What are p-type semi conductors ?
Answer:
Silicon and Germaium when doped with IIIA group elements like B or Al which have only 3 valence electrons. These electrons are bonded to three silicon atoms and fourth valence electron place is vacant. It is called hole. Under the influence of electric field, electrons move towards positive electrode through holes. Hence this type of semi-conductors are called p – type semi conductors.
Question 16.
How many lattice points are there in one unit cell of face – centered tetragonal lattice ?
Answer:
In face centered’tetragonal unit cell
Number of face centered atoms per unit cell
= 6 face centered atoms × \(\frac{1}{2}\) atom per unit cell
6 × \(\frac{1}{2}\) = 3 atoms
∴ Total no. of lattice points = 1 + 3 = 4.
![]()
Question 17.
How many lattice points are there in one unit cell of body centered cubic lattice ?
Answer:
In body – centered cubic unit cell
The number of comer atoms per unit cell
= 8 comers × \(\frac{1}{8}\) per corner atom
= 8 × \(\frac{1}{8}\) = 1 atom
Number of atoms at body center = 1 × 1 = 1 atom
∴ Total no. of lattice points = 1 + 1 = 2.
Short Answer Questions
Question 1.
Calculate the efficiency of packing in case of a metal of simple cubic crystal.
Answer:
Packing efficiency in case of metal of simple cubic crystal:
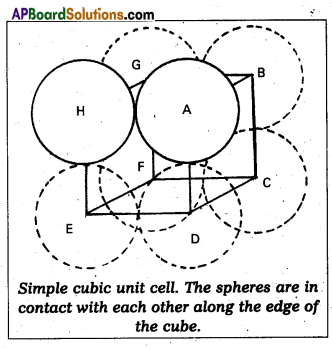
The edge length of the cube
a = 2r. (r = radius of particle)
Volume of the cubic unit cell = a3 = (2r)3
= 8r3
∵ A simple cubic unit cell contains only one atom
The volume of space occupied = \(\frac{4}{3}\) πr3
∴ Packing efficiency
= \(\frac{\text { Volume of one atom }}{\text { Volume of cubic unit cell }}\) × 100
= \(\frac{4 / 3 \pi r^{3}}{8 r^{3}}\) × 100 = \(\frac{\pi}{6}\) × 100 = 52.36%.
Question 2.
Calculate the efficiency of packing in case of a metal of body centered cubic crystal.
Answer:
Packing efficiency in case of a metal of body centred cubic crystal:
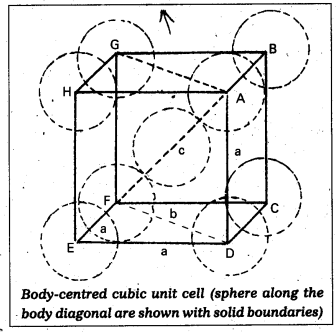
in B.C.C. Crystal
\(\sqrt{3}\)a = 4r
a = \(\frac{4 \mathrm{r}}{\sqrt{3}}\)
In this structure total no. of atoms is ‘2’ and their volume = 2 × (\(\frac{4}{3}\)) πr3
Volume of the cube = a3 = (\(\frac{4}{\sqrt{3}}\)r)3
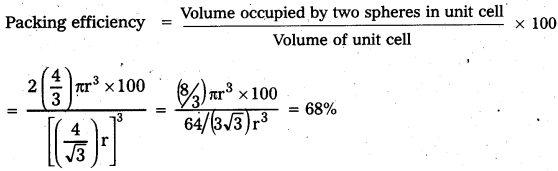
![]()
Question 3.
Describe the two main types of semiconductors and contrast their conduction mechanism.
Answer:
The solids which are having moderate conductivity between insulators and conductors are called semi conductors.
- These have the conductivity range from 10-6 to 104 Ohm-1m-1.
- By doping process the conductivity of semi conductors increases.
E.g.: Si, Ge, crystal.
Semi conductors are of two types. They are :
1. Intrinsic semi-conductors : In case of semi-conductors, the gap between the valence band and conduction band is small. Therefore, some electrons may jump to conduction band and show some conductivity. Electrical conductivity of semi-conductors increases with rise in “temperature”, since more electrons can jump to the conduction band. Substances like silicon and germanium show this type of behaviour and are called intrinsic semi-conductors.
2. Extrinsic semi – conductors : Their conductivity is due to the presence of impurities. They are formed by “doping”.
Doping: Conductivity of semi-conductors is too low to be of pratical use. Their conductivity is increased by adding an appropriate amount of suitable impurity. This process is called “doping”.
Doping can be done with an impurity which is electron rich or electron deficient.
Extrinsic semi-conductors are of two types.
a) n-type semi-conductors : It is obtained by adding trace amount of V group element (P, As, Sb) to pure Si or Ge by doping.
When P, As, Sb (or) Bi is added to Si or Ge, some of the Si or Ge in the crystal are replaced by P or As atoms and four out of five electrons of P or As atom will be used for bonding with Si or Ge atoms while the fifth electron serve to conduct electricity.
b) p-type semi-conductors : It is obtained by doping with impurity atoms containing less electrons i.e., Ill group elements (B, Ai, Ga or In).
When B or AZ is added to pure Si or Ge, some of the Si or Ge in the crystal are replaced by B or AZ atoms and four out of three electrons of. B or AZ atom will be used for bonding with “Si” or Ge atoms while the fourth valence electron is missing is called electron hole (or) electron vacancy. This vacancy on an atom in the structure migrates from one atom to another. Hence it facilitates the electrical conductivity.
![]()
Question 4.
Derive Bragg’s equation. [A.P. & T.S. Mar. 17, 16; IPE Mar & May 15]
Answer:
Derivation of Bragg’s equation: When X-rays are incident on the crystal or plane, they are diffracted from the lattice points (lattice points may be atoms or ions or molecules). In the crystal the lattice points are arranged in regular pattern. When the waves are diffracted from these points, the waves may be constructive or destructive interference.
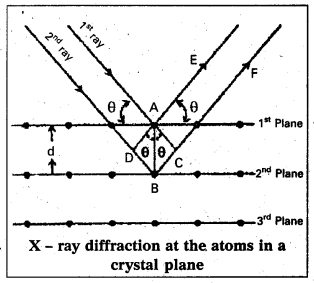
The 1st and 2nd waves reach the crystal surface. They undergo constructive interference. Then from the figure 1st and 2nd rays are parallel waves. So, they travel the same distance till the wave form AD. The second ray travels more than the first by an extra distance (DB + BC) after crossing the grating for it to interfere with the first ray in a constructive manner. Then only they can be in the same phase with one another. If the two waves are to be in phase, the path difference between the two ways must be equal to the wavelength (X) or integral multiple of it (nλ, where n = 1, 2, 3, ………..)
(i.e.,) nλ = (DB + BC) [where n = order of diffraction]
DB = BC = d sin θ [θ = angle of incident beam,]
(DB + BC) = 2d sin θ [d = distance between the planes]
nλ = 2d sin θ
This relation is known as Bragg’s equation.