These AP 9th Class Physical Science Important Questions 3rd Lesson Is Matter Pure? will help students prepare well for the exams.
AP State Syllabus 9th Class Physical Science 3rd Lesson Important Questions and Answers Is Matter Pure?
9th Class Physical Science 3rd Lesson Is Matter Pure? 1 Mark Important Questions and Answers
Question 1.
There is a mixture with sand and iron filings. Write an activity for the separation of iron filings from sand.
Answer:
- Take mixture of sand and iron filing in a tub.
- Take a magnet and move over the mixture.
- Iron filings are separated by sticking to the magnet.
9th Class Physical Science 3rd Lesson Is Matter Pure? 2 Marks Important Questions and Answers
Question 1.
Frame two questions to understand “Homogeneous mixture”.
Answer:
- Can you see components in homogenous mixture?
- In which mixture components are uniformly distributed, heterogeneous or homogeneous?
![]()
Question 2.
How can you use milk to show Tyndall effect?
Answer:
- Take milk in a glass.
- Pass light through the milk.
- Milk particles shine due to scattering of light.
- This is the Tyndall effect.
- Milk is a colloidal solution.
9th Class Physical Science 3rd Lesson Is Matter Pure? 4 Marks Important Questions and Answers
Question 1.
Draw the arrangement of apparatus of fractional distillation experiment. What is the advantage of using the fractionating column?
Answer:
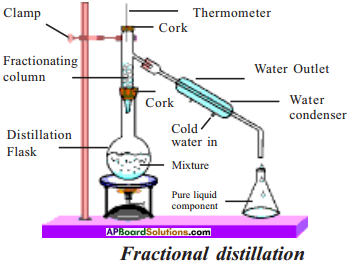
- Fractional distillation column contains glass beads.
- These glass beads in fractional distillation column provide maximum possible surface area for the vapours to cool and condense repeatedly.
Question 2.
Draw and label the apparatus set up for the separation of a mixture by sublimation.
Answer:
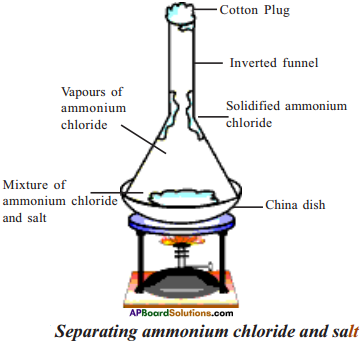
Question 3.
Draw and label the apparatus set up for the separation of a mixture by evaporation.
Answer:
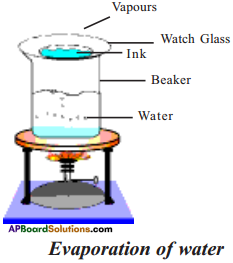
Question 4.
Explain how two miscible liquids can be separated if their boiling points are close to each other.
Answer:

To separate two or more miscible liquids when the difference in their boiling points is less than 25°C, fractional distillation process is used.
Aim :
To separate two miscible liquids (water and acetone) by fractional distillation.
Materials required :
Stand, distillation flask, thermometer, condenser, beaker, acetone and water, one holed rubber cork.
Procedure:
- Take a mixture of acetone and water in a distillation flask.
- Fix a thermometer as shown in the figure and clamp to the stand.
- Attach condenser to the flask.
- Keep a beaker to collect distillate at the end of condenser.
- Heat the mixture slowly.
- Observe the reading of thermometer.
- The acetone (low boiling point) vapourises and condenses.
- It can be collected from the condenser outlet.
- Water remains in the distillation flask. ,
Question 5.
800 ml of solution contains 20 grams of solute. Calculate the concentration in terms of mass by volume percentage of the solution.
Answer:

Question 6.
a) Diagram shows one of the process of separation by mixtures. Based on the diagram answer the following questions.
i) Identify the process involved in the diagram.
Answer:
Sublimation.
ii) Is something missing in the diagram. If so what is that?
Answer:
Stove is missed for heating.
iii) If ‘B’ represent ammonium chloride, then what is ‘A’ represent?
Answer:
Mixture of ammonium chloride and salt.
iv) Give one more example for separation of mixture using above process?
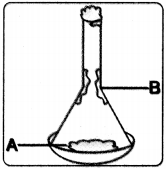
Answer:
Camphor and salt.
Question 7.
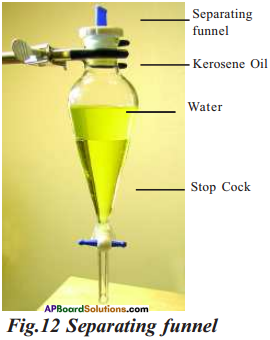
Name the instrument used to separate immiscible liquids. Draw a neat diagram of it taking kerosene and water as immiscible liquids.
Answer:
The instrument used to separate immiscible liquids is separating funnel.
9th Class Physical Science 3rd Lesson Is Matter Pure? Important Questions and Answers
9th Class Physical Science 3rd Lesson Is Matter Pure? 1 Mark Important Questions and Answers
Question 1.
What is meant by ‘Pure substance’?
Answer:
In our day to day language, ‘pure’ means something with no adulteration. A substance is said to be pure i.e., homogeneous when the composition doesn’t change, no matter which part of the substance you take for examination.
Question 2.
What is a mixture?
Answer:
A mixture is generally made of two or more components that are not chemically combined.
![]()
Question 3.
What is a homogeneous mixture? Give examples.
In a homogeneous mixture the components of the mixture of uniformly distributed throughout it.
Ex : Lemonade, sugar solution, air, etc.
Question 4.
What is a heterogeneous mixture? Give examples.
Answer:
A heterogeneous mixture is a mixture made up of different substances or the same substance in different states which are not uniformly distributed in it.
Ex : Mixture of oil and water; water and naphthalene, etc.
Question 5.
What are the factors affecting rate of dissolving?
Answer:
The factors affecting rate of dissolving are
i) Temperature of solvent,
ii) Size of solute particles,
iii) Stirring of the solution.
Question 6.
When do you say that a solution is dilute solution?
Answer:
If the amount of solute present is little, the solution is said to be dilute.
Question 7.
What do you say that a solution is a concentrated solution?
Answer:
If the amount of solute present is more in a solution, then the solution is said to be a concentrated solution.
Question 8.
Define suspension.
Suspensions are the heterogeneous mixtures of a solid and a liquid in which the solids do not dissolve, like mixtures of soil and water.
Ex : Mixture of sand and water.
Question 9.
Define emulsion and give an example.
Answer:
Emulsion is a mixture consisting of two liquids that do not mix and settle into layers when they left undisturbed.
Ex : Mixture of oil and water.
![]()
Question 10.
What is a colloid?
Answer:
Colloids are heterogeneous mixtures in which the particle size is too small to be seen with the naked eye, but big enough to scatter light.
Ex : Milk, Cheese, Ghee, etc.
Question 11.
What is Tyndall effect?
Answer:
Scattering of a beam of light is called Tyndall effect.
Question 12.
What technique do you use to separate the colours?
Answer:
The laboratory technique called chromatography is used for the separation of mixtures into its individual components like inks and dyes.
Question 13.
When do we use fractional distillation method for the separation of miscible liquids?
Answer:
To separate two or more miscible liquids when the difference in their boiling points is less than 25°C, fractional distillation process is used.
Question 14.
What is the definition given by Lavoisier for the ‘element’?
Answer:
According to Lavoisier, an element is a form of matter that cannot be broken down by chemical reactions into simpler substances.
Question 15.
What is the principle involved in separation of immiscible liquids using separation funnel?
Answer:
The underlying principle involved in separation of immiscible liquids using separating funnel is that the immiscible liquids separate out into layers depending on their densities.
Question 16.
What is the use of glass beads in the fractional distillation column?
Answer:
The glass beads in fractional distillation column provide maximum possible surface area for the vapours to cool and condense repeatedly.
Question 17.
How will you separate a mixture containing kerosene and petrol (difference in their boiling points is more then 25°C). Which are miscible with each other?
Answer:
Kerosene and petrol are separated by using distillation process because the difference in boiling point is more than 25°C.
Question 18.
What type of mixtures are separated by the techniques of crystallisation?
Answer:
Salt from water, sugar from water and sodium chloride from its solution in water, etc. are the mixtures to be separated by the techniques of crystallisation.
9th Class Physical Science 3rd Lesson Is Matter Pure? 2 Marks Important Questions and Answers
Question 1.
What is a centrifuge? What are its uses?
Answer:
Centrifuge is a machine used to separate the particles of higher mass and lower mass from a mixture.
Uses :
- To separate cream from milk.
- In diagnostic laboratories, to test blood and urine samples.
- Dryer in a washing machine.
Question 2.
Define a mixture and mention its properties.
Answer:
Mixture :
A mixture is generally made of two or more components that are not chemically combined.
Properties :
- The substances in a mixture retain their own properties.
- The substances in a mixture can be physically separated.
Question 3.
What are homogeneous and heterogeneous mixtures? Give examples.
Answer:
Homogeneous mixture:
A homogeneous mixture is a mixture in which the components of the mixture are uniformly distributed throughout it.
Ex :
Lemonade, sugar solution, etc.
Heterogeneous mixture :
A heterogeneous mixture is a mixture made up of different substances or the same substance in different states which are not uniformly distributed in it.
Ex :
Oil and water; Naphthalene and water, etc.
Question 4.
Define solution, solvent and solute.
Answer:
Solution :
The homogeneous mixture of two or more substances is that we cannot separate them by the process of filtration called a solution.
Solvent:
The component of the solution that dissolve the other component in it (usually the component present in larger quantity) is called solvent.
Solute :
The component of the solution that is dissolved in the solvent (usually the component present in lesser quantity) is called solute.
![]()
Question 5.
Mention the properties of a solution.
Answer:
- The particles of a solution cannot be seen with our naked eye.
- They do not show Tyndall effect.
- The solute particles do not settle down when left undisturbed.
- Solution is a homogeneous mixture.
Question 6.
What are the disperse phase and dispersion medium of a colloidal solution?
Answer:
Disperse phase :
Disperse phase is the substance that present in small proportions and consists of particles of colloidal size (lnm to 100 nm).
Dispersion medium :
Dispersion medium is the medium in which the colloidal particles are dispersed.
Question 7.
Define miscible and immiscible liquids.
Answer:
Miscible liquid :
A liquid is said to be miscible if it dissolve completely in another liquid.
Ex : Alcohol is miscible in water.
Immiscible liquid :
An immiscible liquid is one which doesn’t dissolve but forms a layer over another liquid and can be separated easily.
Ex : Oil is immiscible in water.
Question 8.
Define element and compound. Give examples.
Answer:
Element:
Element can be defined as a basic form of matter that cannot be broken down into simpler substances by chemical reactions.
Ex : lron, gold, silver, sodium, magnesium, etc.
Compound :
Compound can be defined as pure substance that can be separated into two or more components by means of chemical reactions.
Ex : Copper sulphate, ammonium chloride, etc.
Question 9.
How do you appreciate the efforts of scientists in discovering elements?
Answer:
- Generally elements are available in nature in the form of their ores.
- Study of properties of elements lead to the development of civilization.
- The efforts of early alchemists-up to and including Newton, Hennig Brand, Sir Humphry Davy are appreciable for their works in unearthing new elements.
- The definition of element and compound given by Robert Boyle and Lavoisier lead to study the properties of elements and compounds.
Question 10.
Name the technique to separate,
i) butter from curd
ii) salt from sea-water
iii) camphor from salt
Answer:
i) Centrifugation method is used to separate butter from curd .
ii) Crystallisation is used to separate salt from water.
iii) Sublimation technique is used to separate camphor from salt.
Question 11.
What are the factors on which solubility depends on?
Answer:
The factor influence solubility are
- nature of solute
- nature of solvent
- temperature.
Question 12.
What is a concentrated and dilute solution?
Answer:
If the amount of solute is less in a solution, then it is called dilute solution.
If the amount of solute is more in a solution, then the solution is called concentrated solution.
![]()
Question 13.
How do you separate following mixture?
a) Iodine from sodium chloride
b) Petrol from water
c) Butter from milk
d) Sugar from water
Answer:
a) Sublimation
b) Separating funnel
c) Centrifugation
d) Evaporation
Question 14.
Identify pure substance and mixture from this figure. Why?
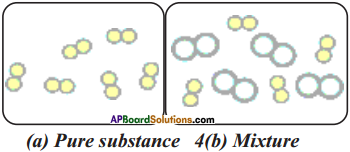
Answer:
a) Fig ‘a’ is a pure substance. Because particles are evenly distributed.
b) Fig ‘b’ is a mixture. Because particles are unevenly distributed.
9th Class Physical Science 3rd Lesson Is Matter Pure? 4 Marks Important Questions and Answers
Question 1.
Define the terms :
a) Solubility
b) Saturated solution
c) Unsaturated solution
d) Concentration
Answer:
a) Solubility :
The amount of solute present in a saturated solution at a certain tem-perature is called its solubility.
b) Saturated solution :
When no more solute can be dissolved in the solution at a certain temperature, it is said to be a saturated solution.
c) Unsaturated solution :
If the amount of solute present in the solution is less than that in the saturated solution is called an unsaturated solution.
d) Concentration of a solution:
The concentration of a solution can be defined as the amount (mass) of solute present in a given amount (mass) of solution or the amount (mass) of solute dissolved in a given volume of the solution.
Question 2.
Compare the properties of suspensions and colloids.
Answer:
| Suspensions | Colloids |
| 1. Suspensions are heterogeneous mixtures. | 1. Colloids are heterogeneous mixtures. |
| 2. The particles of suspensions can be seen with naked eyes. | 2. The size of particles of a colloid are too small to be individually seen by naked eyes. |
| 3. The particles of a suspension scatter a beam of light passing through it and make its path visible. | 3. The particles of colloids are big enough to scatter a of light passing through it which makes its path visible. |
| 4. The solute particles settle down when suspensioin is kept undisturbed. When the particles settle down it does not scatter light any more. | 4. The particles don’t settle down when the colloid left undisturbed, i.e., colloid is quite stable. |
| 5. Suspension is unstable. The components can be separated from the mixture by the process | 5. The components cannot be separated from the mixture by the process of filtration or decantation Centrifugation technique is used in separation. |
Question 3.
Explain the process of preparation of ice cream.
Answer:
- Ice cream is made by churning a mixture of milk, sugar and flavours.
- This mixture is slowly chilled to form ice cream.
- The churning process disperses air bubbles into the mixture by foaming and break up the large ice crystals into tiny particles.
- The result is a complex substance which contains solids, liquids and gases.
- This is the ice cream.
- Ice cream is a colloid.
![]()
Question 4.
What is chromatography? What are its uses?
Answer:
Chromatography :
Chromatography is a laboratory technique for the separation of mixtures into its individual components.
Uses:
- Used to separate components of ink and dyes.
- Used to separate the coloured pigments in plants.
- Used to determine the chemical composition of many substances.
- Used in crime scene investigations.
- Used in hospitals to detect alcohol levels in a patient’s blood stream.
- Used by environmental agencies to detect the level of pollutants in water supplies.
- Used by pharmacists to determine the amount of each chemical found in each product.
Question 5.
Explain the process of separation of components of air briefly.
Answer:
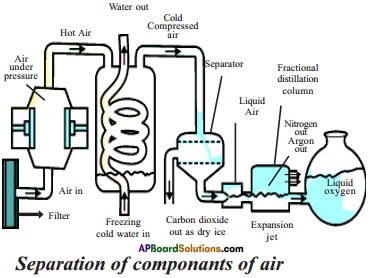
- If we want oxygen gas from air, we have to separate out all the other gases present in air.
- The air is compressed by increasing the pressure and then cooled by decreasing the temperature to get liquid air.
- This liquid air is allowed to warm up slowly in a fractional distillation column where gases get separated at different heights depending upon their boiling points.
Question 6.
Distinguish between mixtures and compounds.
Answer:
| Mixtures | Compounds |
| 1. Elements or compounds just mix together to form a mixture and no new compound is formed. | 1. Elements react to form new compounds. |
| 2. A mixture has a variable composition. | 2. The composition of each new substance is always fixed. |
| 3. A mixture shows the properties of the constituent substances. | 3. The new substance has totally different properties. |
| 4. The constituents can be separated fairly easily by physical methods. | 4. The constituents can be separated only by chemical or electrochemical reactions. |
Question 7.
Draw a flow chart shows the process of obtaining gases from air.
Answer:
- We have learnt that air is a homogeneous mixture.
- It can be separated into its components.
- The following are the steps involved in separating the components of air.

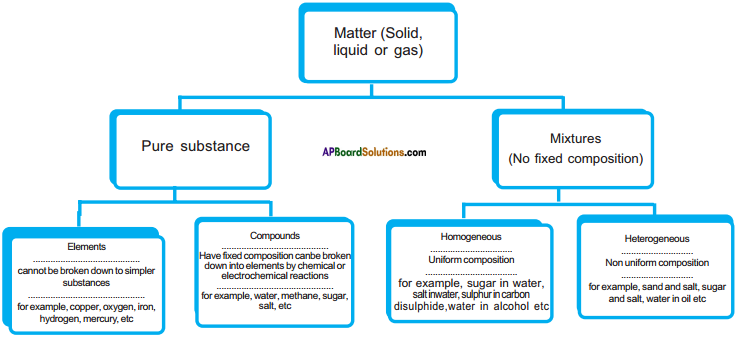
Question 8.
Draw a flow chart to understand the chemical and physical nature of the matter.
Answer:
Question 9.
Give some daily life experiences where you can observe “Tyndall effect”.
(OR)
What is Tyndall effect? Write any two applications of Tyndall effect.
Answer:
Tyndall effect:
Scattering a beam of light is called the ‘Tyndall effect’.
Daily life experiences:
- Select a room where the sun light falls directly through a window.
Close the window in such a way that a slit is left open between the windows.
We observe a beam of light passing through the slit. - While walking on a road having a lot of trees on both sides, when the sun light passes through branches and leaves, we can see the path of dust particles.
- In cinema halls we can observe the beam of light scatters from projector to the screen.
- When smoke in the kitchen is exposed to sun light, we can observe the path of smoke particles.
![]()
Question 10.
How do you separate the mixture of napthaleine powder and salt powder?
Answer:
Aim :
To separate components in the mixture of napthaleine and salt.
Apparatus:
China dish, cotton plug, funnel, burner.
Method :
Separation mixtures by sublimation.
Procedure :
- Take 4 table spoons of mixture in a China dish.
- Take the glass funnel and plug the mouth of the funnel with cotton plug.
- Invert the funnel over the China dish.
- Keep the dish on the stand of stove and heat for some time.
Observation :
- Vapours of napthaleine solidified on the walls of the funnel.
- Salt remains in the China dish.
