These AP 9th Class Biology Important Questions 11th Lesson Bio Geo Chemical Cycles will help students prepare well for the exams.
AP State Syllabus 9th Class Biology 11th Lesson Important Questions and Answers Bio Geo Chemical Cycles
9th Class Biology 11th Lesson Bio Geo Chemical Cycles 1 Mark Important Questions and Answers
Question 1.
What are biogeochemical cycles?
Answer:
The cycles that involve the flow of nutrients in on earth from environment to organisms and back through certain pathways are known as biogeochemical cycles.
Question 2.
Write briefly about assimilation.
Answer:
Nitrogen compounds mainly as nitrates or ammonium ions (NH4+) are taken up from soils by plants which are then used in the formation of plant proteins and as animals eat these plants, animal proteins are synthesised.
![]()
Question 3.
What is bio-geochemical cycle?
Answer:
A constant interaction between biotic and abiotic components of the biosphere makes a system and this flow of components form a cycle is called bio-geochemical cycle.
Question 4.
What is the percentage of nitrogen and oxygen in the air?
Answer:
Nitrogen – 78%
Oxygen-21%
Question 5.
Name few green house gases.
Answer:
Methane (CH4), Carbondioxide (CO2), and Carbon monoxide(CO).
Question 6.
What is denitrification?
Answer:
Conversion of nitrates into free Nitrogen is called denitrification.
Nitrates → Nitrogen(N2)
Question 7.
What is nitrification?
Answer:
Conversion of free nitrogen present in the atmosphere into compounds of nitrogen is called nitrification.
![]()
Question 8.
Name the three processes by which oxygen is lost from the atmosphere.
Answer:
By combustion, respiration, and formation of oxides of nitrogen are the main processes by which oxygen is lost.
Question 9.
Why are root nodules useful for plants?
Answer:
In root nodules, nitrogen fixing bacteria Rhizobium’ is present which fixes the atmospheric nitrogen and make it available to the plant for its growth.
Question 10.
What is acid rain?
Answer:
The gases released due to combustion of fossil fuels are SO2, NO2, CO2. These gases remain suspended in the air. When it rains, the rain water mixes with these gases to form sulphuric acid, nitrous acid, carbonic acid and comes down on the surface of the earth in the form of acid rain.
Question 11.
Give the names of few organisms that help in nitrogen fixation?
Answer:
- Rhizobium helps in the fixation of atmospheric Nitrogen into Ammonia.
- Nitrosomonas converts ammonia to Nitrates
- Nitrobactor converts nitrites into nitrates.
Question 12.
List any three activities which would lead to an increase in the carbon dioxide content of air.
Answer:
- Burning of fossil fuels like coal, petroleum
- Industrialisation
- Deforestation
![]()
Question 13.
Which gas is added to the atmosphere by decay of organic matter?
Answer:
CO2 (Carbondioxide)
Question 14.
List out three recently organized problems of environment.
Answer:
Ozone layer depletion, Green house effect, and Global warming.
Question 15.
What would happen if ail oxygen present in the environment is converted to ozone?
Answer:
Ozone is a poisonous gas and is present in a thin layer in the stratosphere. If all the oxygen is converted to ozone (O3) the environment becomes poisonous and kills all living things.
Question 16.
Expand CFC’s.
Answer:
Chloro Fluoro Carbons
Question 17.
Expand B.O.D.
Answer:
Biological Oxygen Demand
![]()
Question 18.
What is the main concern of montreal protocol?
Answer:
To control the use of gases which have destructive effect on ozone layer.
Question 19.
Define Eutrophication.
Answer:
Increase growth of algae in lakes and ponds is called Eutrophication.
Question 20.
Which ’forms’ of nitrogen are taken up by plants from the soil?
Answer:
Nitrates and Ammonium ions.
Question 21.
What is the most abundant element in the atmosphere?
Answer:
Nitrogen.
Question 22.
What is called as ’Universal Solvent’?
Answer:
Water (H2O).
![]()
Question 23.
How much percentage of water on the earth is present in the ocean?
Answer:
97%
Question 24.
What is the other name for water cycle?
Answer:
Hydrological cycle.
Question 25.
How many oxygen atoms are present in Ozone?
Answer:
Three – (O3).
Question 26.
What is the chemical formula of ammonium ion?
Answer:
NH4+
Question 27.
What are the affects of ‘Global warming’?
Answer:
1) Global warming affects the weather patterns on Earth and causes climatic change.
2) Climatic changes results in higher sea levels, more rainfall, severe droughts and floods.
9th Class Biology 11th Lesson Bio Geo Chemical Cycles 2 Marks Important Questions and Answers
Question 1.
What are the uses of water?
Answer:
- Water participates in many biochemical mechanisms, including photosynthesis, digestion, and cellular respiration.
- Living organisms drink water.
- Water is also used by terrestrial animals and plants for various life processes.
- Water provides hydrogen and oxygen that form integral part of basic organic compounds of life.
Question 2.
What is nitrification? What are the bacteria that participate in nitrification?
Answer:
- Nitrates can also be converted to ammonia by the denitrifying bacteria in the soil.
- The nitrifying bacteria may then use this ammonia to synthesize compounds for their own cell and eventually convert to proteins, nucleic acids, nitrites, and nitrates.
- Nitrites are produced mainly by nitrosomonas, while nitrates by nitrobacters that are also capable of utilizing nitrites and converting them to nitrates.
- Death of these organisms add the nitrogenous compounds to the soil.
![]()
Question 3.
What is ammonification? Under which conditions ammonification occurs?
Answer:
- Production of ammonia (NH3) from nitrates and other nitrogenous compounds is called ammonification.
- Ammonification also occurs when plants and animals die, or when animals emit wastes, the nitrogen in the organic matter reenters the soil and water bodies where it is broken down by other organisms, known as decomposers.
Question 4.
What is denitrification? Explain the process of denitrification.
Answer:
- Denitrification is the conversion, principally by bacteria, of compounds of nitrogen in solid and aquatic systems to the gases, nitrogen (N2) and nitrous oxide (N2O) and eventual release of these into the atmosphere.
- Denitrification occurs primarily in wet soils where water makes it difficult for microorganisms to get oxygen.
- Under these conditions, certain organisms – known as denitrifying bacteria – will process nitrate to gain oxygen, leaving free nitrogen gas as by-product.
Question 5.
Explain briefly about green house effect.
Answer:
- Some gases present in the atmosphere such as carbon dioxide, carbon monoxide, methane, and water vapour trap heat from radiating back to the space.
- The natural green house gases act like a big blanket around the earth, keeping it warm and making life possible without which temperatures would have fallen to sub zero values.
- This phenomenon of naturally warming up is called ‘Green house effect’.
Question 6.
What are the uses of oxygen?
Answer:
- Oxygen helps in burning things.
- Oxygen is required for survival of living organisms.
- Respiration utilizes oxygen releasing carbon dioxide to atmospheric pool maintaining a balance in nature.
- Dissolved oxygen supports aquatic life.
- Oxygen is needed for the decomposition of organic waste.
![]()
Question 7.
What is biological oxygen demand? Explain briefly.
Answer:
- The content of biodegradable substances in water is expressed by a special index called “biological oxygen demand” (BOD).
- BOD represents the amount of oxygen needed by aerobic bacteria to decompose the waste.
- As the wastes get degraded and the dissolved oxygen is used up proportionately, the need or demand for oxygen increases i.e. the BOD increases.
- Thus BOD is a good indicator for amount of biodegradable waste.
Question 8.
What are the differences between oxygen and ozone?
Answer:
- Oxygen has two atoms and it is colorless and odourless.
- Ozone is a molecule containing three oxygen atoms. It is blue in colour and has a strong odour.
Question 9.
Complete the following flow chart.
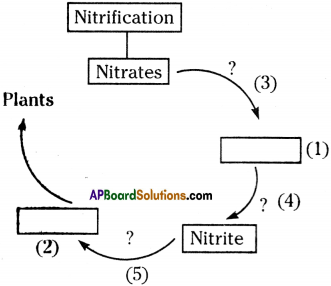
Answer:
- Ammonia
- Nitrate
- Denitrifying bacteria
- Nitrosomonas
- Nitrobactor
Question 10.
How is a balance maintained in the environment?
Answer:
The living or biotic components (plants and animals) and the non – living or physial components (air, water, soil, light and temperature) interact and affect each other resulting in the establishment of a complex and complete balance in the environment. There is a continuous cycle of nutrients among the biotic and abiotic components.
Question 11.
Write the two harmful effects of ozone hole.
Answer:
- Ozone hole allows harmful U.V radiation to reach the earth’s surface that may cause skin cancer, cataract, etc
- Ultraviolet rays also ionize the cells, change the gene structure in animals and plants.
Question 12.
What are the harmful effects of acid rain?
Answer:
- It makes the soil acidic.
- It damages the crops and plantations.
- It corrodes statues, monuments of marble buildings, etc…
![]()
Question 13.
What is the difference in fog and smog? Give two harmful effects of smog.
Answer:
The water vapour present in air when condenses due to very low temperature is called fog. The smoke released in the air due to burning of fuels mixes with the fog and forms smog.
Smoke + Fog = Smog
a) Smog reduces visibility
b) Smog creates respiratory diseases like asthma etc.
Question 14.
What is bad ozone and good ozone?
Answer:
Ozone when present in the earth’s surface is very poisonous gas hence called bad ozone. When ozone present in stratosphere is very protective in nature as it does not allow the harmful U.V radiation to enter the earth’s atmosphere and hence is called good ozone.
Question 15.
Consider that there is a mass mortality of Ashes in a pond. Guess, what may be the reason for that.
Answer:
- Addition of hot water to the fish tank may leads to their death.
- Addition of poisonous mercury compounds in water may leads to their death.
- Blockage of gills of fishes with any pollutant may be .a cause for their mass death.
![]()
Question 16.
Why is it said that nitrogen is important for us?
Answer:
- Nitrogen is the most abundant gas in our atmosphere (78%)
- Nitrogen is a part of many molecules essential to our life like proteins, nucleic acids, (DNA and RNA), and some vitamins.
- It is found in other biologically important compounds also such as alkaloids and urea.
9th Class Biology 11th Lesson Bio Geo Chemical Cycles 4 Marks Important Questions and Answers
Question 1.
Write about the expansion of water on earth.
Answer:
- Nearly 97% of all the water on the earth is in the oceans, and so only about 3% is freshwater.
- About 2% of this fresh water is permanently frozen in glaciers and at the polar ice caps.
- Thus only about 1% is available fresh water.
- Again about 1/4 of this 1% is present as underground water.
- Only about 0.009% of water on earth is in the rivers and lakes.
- Rest is present in the bodies of living organisms, as soil moisture, as humidity of atmosphere, etc.
- Water is the most essential, abundant substance in living things.
- The human body, for example, is composed of about 70% of water.
Question 2.
What is water cycle? Explain it briefly.
Answer:
- The whole process in which water evaporates and falls back on the surface of the earth as rain and other forms of precipitation including its flow from land into the sea/oceans via several routes like rivers, ground water channels, etc., is known as the water – cycle.
- All of the water that falls on the land does not immediately flow back into the sea.
- Some of it seeps into the soil and becomes part of the underground reservoir of fresh water.
- Some of this underground water finds its way to the surface through springs.
- We bring it to the surface for our use through wells or tube wells.
- Water is continuously exchanged between sea, land and atmosphere.
Question 3.
What is nitrogen fixation? How it occurs?
Answer:
- Atmospheric nitrogen occurs primarily in inert form (N2) or non-reactive form that few organisms can use; therefore it must be converted into a compound (or) fixed form in a process called nitrogen fixation.
- Most atmospheric nitrogen is ‘fixed’ through biological processes.
- A number of bacteria and blue green algae are known to be able to fix atmospheric nitrogen into compounds in their own body.
- These may be symbiotic e.g.: Rhizobium or free living, e.g.: Nitrosomonas respectively.
- These organisms convert atmospheric nitrogen into the organic nitrogen for their own cells.
- In leguminous plants like pea, beans, etc., there is a symbiotic relationship of the nitrogen fixing bacteria with the plant, thus nitrogenous compounds are added to the soil after a leguminous crop is grown.
- Nitrogen can also be fixed as nitrates by lightning.
![]()
Question 4.
In which forms carbon found on earth?
Answer:
- Carbon is found in various forms on the earth.
- It occurs in the elemental form as say soot, diamond, and graphite.
- In the combined state, it is found as gases, carbon dioxide, and carbon monoxide in the atmosphere, as carbonate it is found in various salts and minerals.
- While all life – forms are composed of carbon containing molecules like proteins, carbohydrates, fats, nucleic acids, and vitamins.
- The endoskeletons and exoskeletons of various animals are also formed from carbonate salts.
Question 5.
What is Global warming? What are its effects?
Answer:
- Due to human activities, an excessive amounts of carbon dioxide and other green house gases has been emitted to the environment.
- As a result, more heat gets trapped.
- This causes the temperature of the earth to rise, which results in global warming.
- Global warming is the recorded increase in the average temperatures of the earth’s atmosphere and oceans.
- Global warming affects the weather patterns on earth and cause climate change.
- Climate change results in higher sea levels, more rainfall, and severe droughts and floods.
Question 6.
Describe carbon cycle briefly.
Answer:
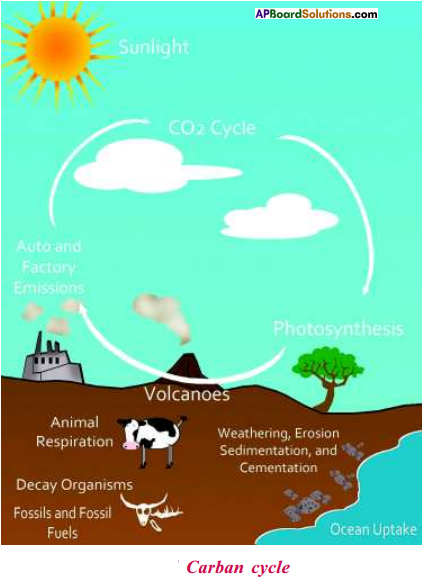
- Carbondioxide is used by plants for photosynthesis.
- During this process, CO2 is converted to glucose.
- Some amount of this glucose is used for respiration by plant.
- Rest of it converted to other carbohydrates and fats stored in various parts of plants.
- When animals eat plants these carbon containing compounds are taken up by the animals and digested and absorbed.
- These are used by animals for respiration.
- Thus carbon in carbon dioxides in atmos-phere finds its way into animals.
- Metabolic degradation of these compounds especially during respiration releases the CO2 into air.
- Decomposition of dead bodies of animals and plants by bacteria also releases CO2 into air.
- Thus the CO2 find its own way into the bodies of animals and plants and back to air.
Question 7.
Briefly describe oxygen cycle.
Answer:
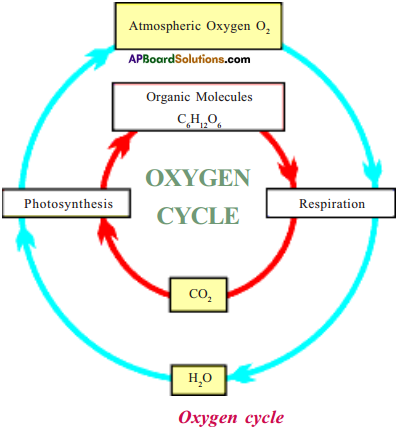
- Oxygen dissolved in water is used for respiration by aquatic plants and animals,
- Terrestrial animals use oxygen present in air for their respiration.
- The by-products of respiration, CO2 and water are released into air.
- These two are taken up by plants and are used for photosynthesis.
- The by-product of photosynthesis is oxygen, which is released into air.
- CO2 is also formed when fuels are burnt.
- There is fine balance between these processes so that the total amount of oxygen present in air remains constant.
- All these processes together represent oxygen cycle.
9th Class Biology 11th Lesson Bio Geo Chemical Cycles Important Questions and Answers
Question 1.
Asa responsible citizen what measures you would follow to reduce global warming at your school or village level?
Answer:
- Planting trees in open fields and wastelands.
- Minimising the usage of motorcycles and start using cycles which are eco-friendly.
- Stop burning of firewood, which release the green house gases start using LPG cylinders for cooking.
![]()
Question 2.
What is nitrification? Explain how it occurs.
Answer:
- The denitrifying bacteria present in the soil convert the nitrates into Ammonia.
- Nitrifying bacteria utilises this ammonia and form proteins, nitrites, nitrates.
- Nitrosomonas produce nitrites whereas nitrobactor produce nitrates.
- Due to the death of microorganisms in the soil, the soil becomes nitrogen rich.
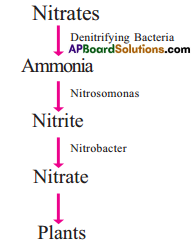
Question 3.
Write differences between nitrification and denitrification.
Answer:
| Nitrification | Denitrification |
| 1) The part of nitrogen cycle, whereammonium (NH4+) is converted into Nitrate (NO3–) is called nitrification. | 1) This is the level where reduction of nitrate (NO3–) is made into Nitrogen (N2) |
| 2) This involves nitrifying bacteria like Nitrobactor Nitrosomonas. | 2) This involves denitrifying bacteria like 4 spirillum, lacto bacillus, pseudomonas thiobacillus. |
| 3) Occurs slowly. | 3) Occurs rapidly. |
| 4) This requires aerobic condition. | 4) This requires anaerobic condition. |
| 5) The microbes involved in this process are autotrophs. | 5) The microbes involved in this process are heterotrophs. |
| 6) The end product is nitrate. | 6) The end product is nitrogen. |
Question 4.
Explain how human activities lead to global warmig.
Answer:
- Deforestation
- Burning of excess of fossil fuels in industries and automobiles.
- Power genaration and utilization
- Refrigerators, coolers, cell towers
- Automobiles and cement roads etc.
- Forest fires and burning of firewood.
Question 5.
Observe the following figure and answer the following questions.
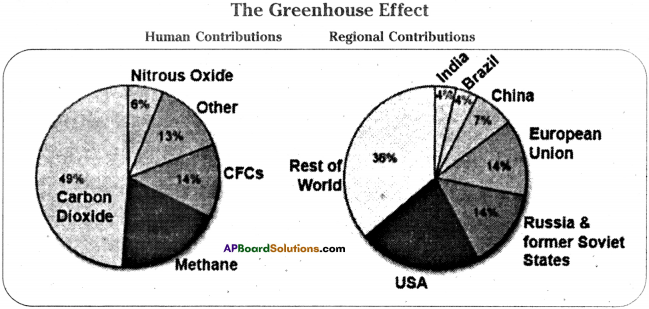
a) What is green house effect?
b) Apart from rest of the world which country contributes more for it?
c) What are CFCs? Give some examples.
d) Suggest some preventive measures to reduce it.
Answer:
a) The phenomenon of naturally warming up by CO2 and water vapour is called “Green house effect”.
b) USA
c) Fully or partly halogenated paraffin hydro carbons that contain only carbon (C), hydrogen (H) chlorine (Cl) and fluorine (F) which are volatile are called chloro fluorine.
Ex : Freon – 12, R – 410A, R -134
d) i) Stop deforestation
ii) Restrict the usage of fossil fuels
iii) Introducing eco friendly fuels like CNG.
iv) Introducing alternative energy resources like solar energy, wind power etc.
